The largest controlled study of psilocybin – the psychoactive ingredient in magic mushrooms – has found the compound safe for human consumption, bringing researchers one step closer to developing a psilocybin-based treatment for depression.
Volunteers who received doses of the psychedelic compound experienced no serious adverse effects in phase one clinical trials at Kings College London (KCL).
Psilocybin has been tipped as a potentially groundbreaking treatment for mental health disorders that could replace antidepressants, with some research suggesting it could also aid those dealing with addiction.
Most of the minor adverse events recorded were of the expected psychedelic nature, researchers found, with changes to sensory perception and mood, but no negative effects on cognitive and emotional functioning.
Research by the company behind the trial, Compass Pathways, into using psilocybin as a treatment for depression has been fast-tracked in the US, receiving special “breakthrough therapy” status from the Food and Drug Administration.
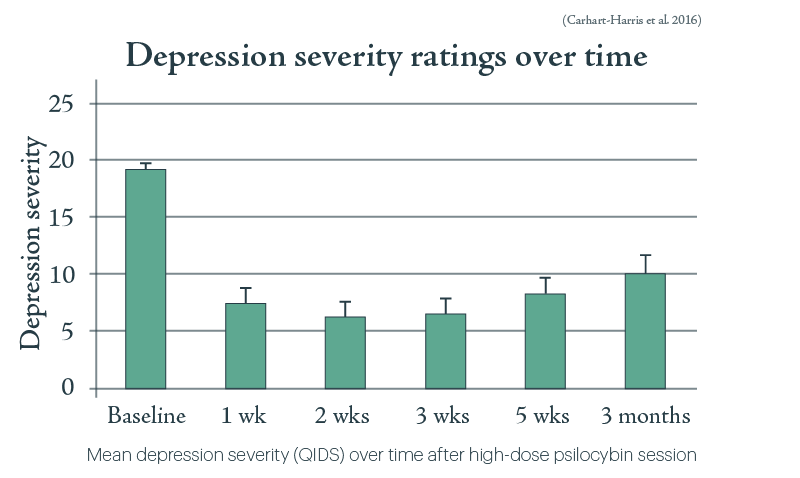
As the study’s lead Dr Robin Carhart-Harris pointed out, this is the polar opposite of antidepressants, which patients often complain leave their emotions “blunted”.
Microdosing
Microdosing involves taking roughly one-tenth the “trip” dose of a psychedelic drug, an amount too little to trigger hallucinations but enough, its proponents say, to sharpen the mind. Psilocybin microdosers report that the mushrooms can increase creativity, calm anxiety, decrease the need for caffeine, and reduce depression.Note:
We are not selling mushrooms, but we known of a trusted source who helps people around uk for a long time, please contact magic@shroomzy.co.uk for details.
According to some researches, magic mushrooms are magical fungi because there are already hundreds of people who are suffering from mental disorders like anxiety, and severe depression is already cured. So, there are great possibilities that this magic mushrooms will be a full treatment in the future.

